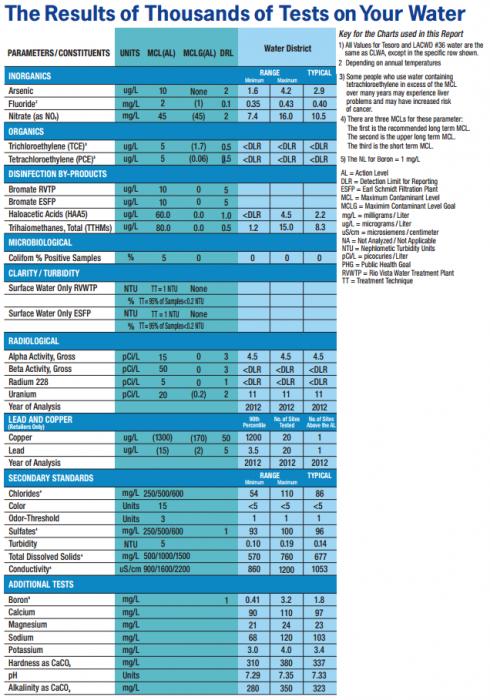
Oh, AJ! There you go off on a minutia rant again. Sure bicarbonate is not directly measured, but it is calculated via alkalinity and pH measurements.
Somebody must be having a bad day. The man asked if bicarbonate was hidden in the report somewhere or whether a conversion was necessary. I told him that it isn't measured but can be estimated and how that is usually done pointing out the weaknesses of the usual simple conversion.
You should go on to say that many other ions are not directly measured, but are actually calculated via surrogates that imply a certain ion concentration.
We measure nothing directly. We always look for a chemical or electrochemical or physical reaction that is
specific (to the extent we can make it so) to one ion for example the response of an ion selective electrode to sodium or nitrate, the absorption or emission of a particular wavelength of light unique to calcium or magnesium (ICP, AAS) or the complexation of mercury or silver to form a colored dye when mercury or silver is no longer consumed by the chloride in a sample. While all these methods are subject to interference we have methods for dealing with them.
There are two cases (that I can think of at the moment) where a brewer does a non specific test and they are alkalinity and total hardness. Total hardness is not particularly useful and so we render the hardness test specific by additions that make it respond only to calcium or magnesium giving us calcium hardness and magnesium hardness as ion specific tests (or test for these with ICP or AAS) leaving alkalinity as the only non specific test.
In alkalinity testing there is no pretense that we are being ion specific. Any base (sulfate, hydroxyl, bicarbonate, carbonate, any of the phosphates, water - yes water has an alkalinity of about 2.5 ppm as CaCO3) react. There is nothing I can do to make the test not respond to any base in the water. The good news is that in the first place bicarbonate is usually the major contributor to alkalinity and in the second that we don't really care what the bicarbonate is (exception: in doing a charge balance computation for analysis quality control). Alkalinity isn't a useful test because it can be used to calculate bicarbonate but because it is a measure of what we really need to know in an acid/base reaction: the proton deficit which must be dealt with in moving a mash or the water to a desired pH.
Oh my god, what will we do now???!!! ;-)
Homebrew and a Prozac?
As AJ finally alludes to:
In the second line of a 4 line post.
the bicarbonate content can be directly calculated from the alkalinity
OK, I've got well water with pH 5.5 and alkalinity of 29.9. What's the bicarbonate content of this water? The answer is 30.4. But if I plug those numbers into the usual formula I come up with 36.4 mg/L. If I do a more careful computation I get 35.9.
Now I've got well water with pH 5.5 and alkalinity of 25.8 and bicarbonate content of 30.7. The formula says it 29.1. Close enough in this case but not so good in the other.
What's the difference here? The biological report on my new well was shocking! That is, it showed that I need to shock the well. For hypochlorite to be effective the pH needs to be reduced and so I've got to put acid in there before dumping in the bleach. If I do this with hydrochloric acid then the bicarbonate simply calculated from the alkalinity and pH is pretty close. If I use phosphoric, then I get almost 20% error. Does an error of this magnitude come under 'minutiae'? I'll leave that to the reader to decide.
When dealing with a water sample which is devoid of phosphate, of course, this doesn't make much difference. But when phosphoric acid is added to it to ajdust pH for sparging or where it is added to malt (which contains phosphate and other bases as well) the relationship between alkalinity and bicarbonate is blown away. One can still do calculations but he had better attend to these minutiae.