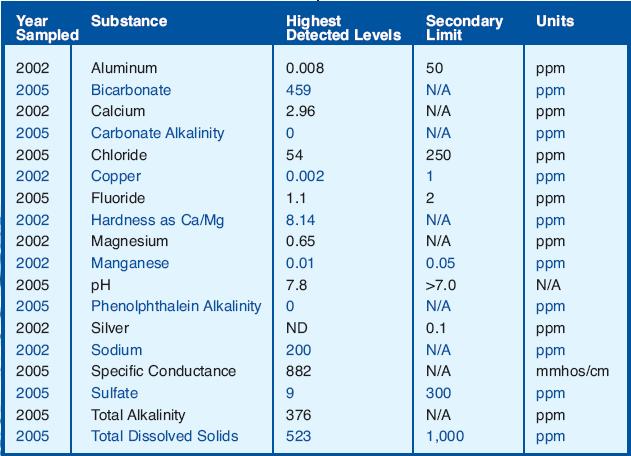
Unless you have particularly hard water or are trying to match a particular water style, you really don't need to worry that much about pH. With regards only to starch conversion, A couple of points:
RO/distilled water has NO real pH!!!!!!! pH is the H+ ion concentration - which there is none of in truely pure water. A pH meter will still give you a reading, and so will various pH papers but they are not giving a true reading. In Theory it should be 7.0 as the concentrations of H+ and OH- are equal (at 0).
Now add a tiny bit of anything with an H+ or OH- and you will get an accurate reading. Adding NaCl for instance will not affect the pH, nor will CaCl2, but CaSO4 will (Sulfate "reacts" with water to give H+ ions). You can add a couple drops of acid and the pH will drop very quickly, but if the concentration is low, it does not take very much of a base to bring it back up. In this case there is very little BUFFERING CAPACITY. THIS is what one needs to pay attention to, not the actual pH. Soft water has little buffering capacity and has the least affect on mash pH. Hard water has a greater buffering capacity and can cause some problems if the water is very hard.
Assuming you have typical water, the pH of the mash is mostly due to the grains you've added, not the water (unless you mash with a high water to grain ratio). The malts are loaded with organic acids, and these are largely responsible for the mash pH. Fully modified malts have a pretty high buffering capacity. Remember the seed is using the same enzymes we're taking advantage of for brewing, to breakdown starch to feed the growing shoots and roots. The seed has set up the components to give it the best pH for it's own starch metabolism (like the acid in our stomach) and we just take advantage of that.
If you fly sparge, as you drain off the wort, you are also draining off the organic acids that are buffering the mash at the proper pH. In this case, towards the end of the sparge your water will have a significant affect on mash pH when the natural organic acid levels go below that of the buffering capacity of your water. If you have problematic water, this is where I'd worry about adjusting pH.
For Batch spargers this is much less of a concern as you aren't contiunally diluting the organic acids.
There are styles where the mash pH should be adjusted to get the proper taste (ie. Berliner Weisse), but I think for most homebrewers, for most beer styles, this is not a big problem.
Of course the water has a big affect on the final taste, but this has more to do with the salts, and not the pH of the mash and starch conversion. The salts in the water have a big impact on hop flavors
From Five Star, the makers of 5.2,
Optimizing the enzymatic activity of your malt
Helping clarify your wort
Obtaining more consistent hop usage in the boil
Reducing scaling & mineral deposition in all your equipment
Brewing more consistent beer!
I've been messing around with this stuff, but not for the first reason listed. My water is a bit high in carbonates and when I added 5.2 to my water and heat it up, it turns cloudy. It is a known fact the Ca will react with phosphate (5.2's main component) to form an insoluble salt so I believe this is why it is cloudy. I "think" also that this may mess up the carbonate equilibrium and result in lower carbonate levels. I need to do more reading to confirm this. I'll know the results in another week what the affect of my experiments with 5.2 are.
 Should I instead perform an extensive acid rest? If so, how long?
Should I instead perform an extensive acid rest? If so, how long?

 . I have very soft water so I can get my pH in the 5.2 - 5.4 range with as little as a 1/4 teaspoon of baking soda. What salts to add and how much, depends on what your water has in it, so check out the information in his book and do the calculation.
. I have very soft water so I can get my pH in the 5.2 - 5.4 range with as little as a 1/4 teaspoon of baking soda. What salts to add and how much, depends on what your water has in it, so check out the information in his book and do the calculation.




























![Craft A Brew - Safale S-04 Dry Yeast - Fermentis - English Ale Dry Yeast - For English and American Ales and Hard Apple Ciders - Ingredients for Home Brewing - Beer Making Supplies - [1 Pack]](https://m.media-amazon.com/images/I/41fVGNh6JfL._SL500_.jpg)