A friend who swears by pH-5.2 says that it helps acidify or alkanize his mash and sparge water.
We have observed that 5.2 works very well for people who do not own pH meters but doesn't work at all for those who do.
I'm no expert but my understanding is that it acts as a buffer for mash water. In other words you add the pH-5.2 to the mash water. You then add your grain and pH-5.2 buffers the acidity of the added grain at a pH of 5.2 such that it doesn't drop below 5.2 unless you exceed the buffering capability of pH-5.2.
Let me start by pointing out that this has been discussed at length on this forum and a search of the archives should turn up a lot of posts.
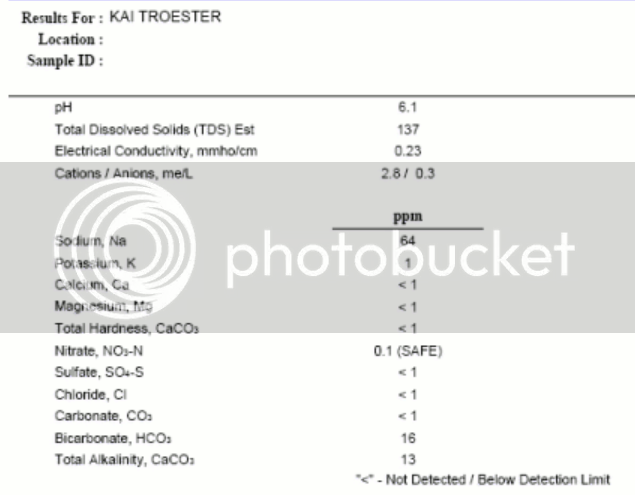
This product is a combination of mono and dibasic sodium phosphate. It is mostly the former. The mix forms a buffer at pH 5.9 (that's not a typo). Remember that malt contains a lot of inorganic phosphate. I am guessing that the idea is to have the sum of the malt monobasic phosphate and the product's monobasic phosphate be such that its ratio to the dibasic (from the product) is such as to have it buffer at 5.2. That may or may not be how they designed it but, as I have stated here and elsewhere dozens of times, you do not design a phosphate buffer for pH 5.2 because that value is too far from either of the spanning pK's of phosphoric acid and, as a consequence, there will be little buffering capacity at pH 5.2. That is why this product comes no where near 'locking on' your pH at 5.2 at normal or even several time the normal dose. The theory says this; experiments in the lab confirm it.
As to the acid base nature of it: at pH 5.2 phosphoric acid is a fairly 'strong' acid. At pH 5.2 dibasic phosphate is a fairly 'strong' base of about equal strength. Thus they tend to neutralize one another with the result being the salt monobasic phosphate. At pH 5.2 there is relatively little phosphoric and relatively little dibasic phophate. The ratio of monobasic ion to phosphoric is pretty high as is the ratio of monobasic to dibasic. There isn't much buffering. At pH 2.1, OTOH, there are equal amounts of phosphoric acid and monobasic ion and at pH 7.2 there are equal amounts of monobasic and dibasic ions. These are the values at which the system will have maximum buffering capacity. The point being that a buffer consists of a mixture of an acid and a base. Keep in mind that the monobasic ion is an acid becase it can give up protons thus lowering pH.
Put another way one could construct a buffer for a pH of say 5.4 or 5.8, etc...
Yes, and they would be better buffers than one designed for 5.2 because 5.4 and 5.6 are closter to 7.2.
I also don't think you would use pH-5.2 in sparge water because what exactly would you be buffering?
A buffer doesn't attack something and in doing so 'buffer' it. Quite the opposite its job is to resist stresses that would result in a pH shift away from its design pH. It absorbs acid/base shocks. Check the Wikipedia article on railroad buffers. Given that your sparge water has no phosphate in it 5.2 will tend to buffer it to around pH 5.9.
IMO, you'd be better off using acid in the sparge water.
If you need to deal with alkalinity and don't object to adding phosphate you will be better off with phosphoric acid than this product because you won't be adding any sodium.
Anyway it's not a water treatment that "snaps" your pH to 5.2, it's simply a buffer at a pH of 5.2 because that's how it was engineered.
It is, in water, designed to buffer to around pH 6. This was confirmed by calculating the ratios of mono and di basic salts, calculating the amount of sodium necessary to balance that and then checking the sodium content of a solution of it.
Am I correct in saying that it doesn't acidify or alkanize your mash? You would need actual water treatment salts for that (Lactic/Citric Acid, CaSO4, CaCl, ...)
If you add monbasic sodium phosphate to any solution or mash which has a pH higher than 4.65 (half way between the first two pK's of phosphoric acid) it will act as an acid. It will give up protons and in the process convert to the dibasic ion. In this sense 5.2, which is mostly the monobasic salt, is an acid and does acidify your mash. If you add a 'real' acid you get the same effect (but the numbers are different). If that acid is phosphoric acid you wind up with the same result: lower pH and a mix of phosphoric acid, monobasic ion and dibasic ion. If adding acid and adding the monobasic salt bring you to the same pH the ratios of the concentrations of the species will be the same.