This is a decent rule of thumb, but it's not exactly 1/4 equivalence... The pressure on a regulator gauge is zeroed at 1 atmosphere, meaning that if your gauge reads 0.1 it is actually already applying 14.8 (1 atmosphere + .1) pounds of pressure. 1/4 of the gas "below" the 0 point in the gauge is also CO2, so you have to account for that in the calculation.
Point being, if you're looking for 2.27 volumes CO2 (12psig at 40º on pure CO2) and serving on beer gas, you're going to end up with less and less dissolved CO2 over the course of the keg. The gas equivalences can be found with these formulas, though:
To find the equivalent pressure for beer gas to give the same CO2 content as pure CO2:
P
beergas = (P
co2 +14.7) / (% CO2 in Mix) - 14.7
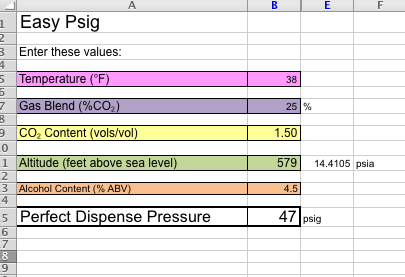
E.g., a mix of 25%CO2/75% N2 would give the same CO2 pressure as 12 psig of pure CO2 at:
= (12+14.7)/(.25) -14.7
= 92.1 psig of beergas
*note: psig = psi of gauge, v. psia = psi absolute
And to find out how much applied pressure the CO2 in a mixed gas is applying on the beer, just reverse it (the 47psi is from your beergas setting):
Papplied = (% CO2 in Mix) * (P
beergas +14.7) - 14.7
= .25 * (47 + 14.7) - 14.7
= 0.725 psig of CO2
You'll notice that under the serving pressures for beer gas, 30 to 45 psig works out to -3.525 psig (or 11.18 psia) of CO2 to 0.225 psig (14.92 psia) of applied CO2. At 40º, this works out to 0.83 to 1.18 volumes of CO2. The fact you can have less than one atmosphere of applied CO2 pressure with beer gas is why you can get these low volumes - at the same temperature, 0.1 psig (just barely moving the needle) on a pure CO2 gas will only get you down to 1.17 volumes CO2.
TL/DR version: Set your beer gas to 30-40 psig and you'll end up with between .8 and 1.2 volumes of CO2. Don't expect to get the same dissolved CO2 as a pure gas unless you really pump up the applied pressure, though...






































![Craft A Brew - Safale S-04 Dry Yeast - Fermentis - English Ale Dry Yeast - For English and American Ales and Hard Apple Ciders - Ingredients for Home Brewing - Beer Making Supplies - [1 Pack]](https://m.media-amazon.com/images/I/41fVGNh6JfL._SL500_.jpg)