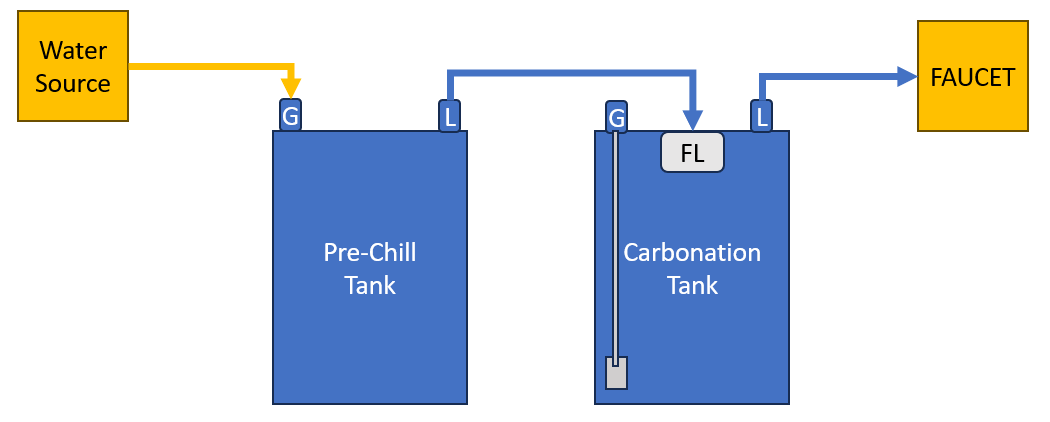
I have served carbonated water from my keezer on and off for a few years. A keg typically lasts a week and it’s then up to me to pull it out and refill the water. Because I can get busy, sometimes we can go a week or more without carbonated water to the dismay of my family. To rectify this issue, I purchased a KegLand continuous soda carbonation keg lid along with a new 9.5 L (2.5 gallon) corny keg. The continuous soda carbonation keg lid has a ball connector on the lid for a water input, that is turned on and off by a float. Thus, as carbonated water is poured, filtered tap water enters which is carbonated via a gas line with a carbonation stone. The idea with the 9.5 L corny keg, is that because it would be constantly refilled, I wouldn’t need a full keg and thus could put the half-sized keg up on the keezer hump, freeing room for a beer keg on the floor.
The issue I am having is that although the pressure on my CO₂ gas line has not changed, I received feedback that the water is not as carbonated as it was before. I am currently on travel but when I get home I am certain I’ll be able to solve the issue by experimenting with increased gas pressure.
My question for the community is, is there some physics going on with the keg having both a pressurized gas and water input? I had assumed that once the water had filled, the tank would look identical to a standard keg and the carbonation stone would deliver the same quantity of gas to the liquid. Is this not the case?
The issue I am having is that although the pressure on my CO₂ gas line has not changed, I received feedback that the water is not as carbonated as it was before. I am currently on travel but when I get home I am certain I’ll be able to solve the issue by experimenting with increased gas pressure.
My question for the community is, is there some physics going on with the keg having both a pressurized gas and water input? I had assumed that once the water had filled, the tank would look identical to a standard keg and the carbonation stone would deliver the same quantity of gas to the liquid. Is this not the case?