jescholler
Well-Known Member
Sodium/Sulfate Harsh Bitterness Experiment
Today I started an experiment to try and determine the cause of a harsh bitterness in my extract beers. At the moment, the experiment is in progress, but I have some good data to present. I plan to update this post as I get information such as fermentation times and temperatures and bottling details. I will add future tasting results in a subsequent post. I hope you find value in this experiment.
Statement of Problem
It is a well known fact that the combination of high levels of sodium and sulfate in brewing water will result in a harsh bitterness in a beer. According to John Palmer, it is desirable to keep either one of these minerals as low as possible, preferably the sodium. The extract brewer is faced with a handicap unless they use water nearly void of all minerals in their beer (i.e. distilled, de-ionized, or put through reverse osmosis). If an extract brewer uses their tap or well water for beer, the total minerals in the final product come from both the water used to make the extract, and the water out of their tap or well. If this sum includes high levels of sodium in combination with sulfate, the beer will have a harsh bitterness.
This was observed in one of my extract brews where I used one gallon of distilled water and the remaining water was from my tap. The initial taste of the beer was great, but it had a harsh bitterness that lasted for many minutes. Upon investigation, I realized that my tap water is high in sodium and sulfate content, 200ppm and 160ppm respectively. Since I was using extract, the minerals in the water used to make the extract also ended up in the final beer. The sum of the sodium and sulfate in both the extract water and my tap water put the harsh bitterness over the top and made the beer unpleasant.
Hypothesis
A simple solution to the problem is to use all distilled water when using extract. This will minimize the amount of both sodium and sulfate in the beer. An extract beer brewed with distilled water will be much less harsh than a beer brewed using my tap water.
Recipe
This single recipe represents two different beers. Both beers are identical to each other except for the water used.
Beer:
Sodium/Sulfate Experiment
Type: Extract
Size: ~.5 gallons
Color: 7 HCU (~6 SRM)
Bitterness: 33 IBU
OG: 1.045
FG: Not Measured (batch too small)
Alcohol: Not Measured (batch too small)
Water:
One beer was made with tap water and 1/8 tsp. gypsum creating the following concentration of minerals:
Calcium - 41ppm
Magnesium - 3ppm
Sodium - 199ppm
*Chloride - 6ppm
Sulfate - 236ppm
The final beer will include the minerals above plus the minerals used in the water to make the extract. It is expected that the combined sodium and sulfate concentrations are extremely high.
*Note the chloride/sulfate ratio of my tap water is extremely weighted to the sulfate side. I have no way of knowing what the mineral content is in the extract I used. I will be able to adjust for the low chloride level by adding calcium chloride to the beer during tasting. If the harsh bitterness is due to the combination of high levels of sodium and sulfate, then adding chloride will not get rid of the harsh bitterness.
The other beer was made with all distilled water, which theoretically has no mineral content. This allows the beer to be made with only the mineral content of the extract.
Boil:
60 minutes
SG: 1.022
1 gallons
8 oz. Amber dry malt extract
Hops:
5g Hallertauer (3.9% AA, 60 min.)
Yeast:
Safale US-05 was used. It was re-hydrated in 1/2 cup distilled water at 80°F. The yeast was allowed to hydrate for 20 minutes and then aerated for the following 30 minutes by swirling occasionally. Each beer received 1 tbsp. of the yeast slurry, equaling approximately 28 billion cells. The yeast was pitched with the worts at ~70°F.
Log:
Brew Date: 7/19/2009
Fermented in 64 oz. growlers at 64°F for 1 month.
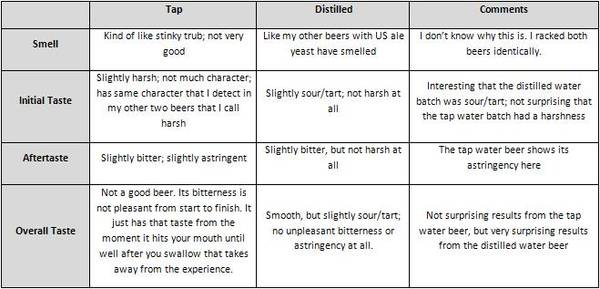
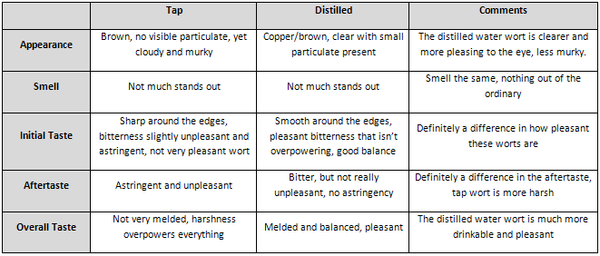
Results
The two worts were sampled prior to pitching the yeast. A comparison is shown below.
Today I started an experiment to try and determine the cause of a harsh bitterness in my extract beers. At the moment, the experiment is in progress, but I have some good data to present. I plan to update this post as I get information such as fermentation times and temperatures and bottling details. I will add future tasting results in a subsequent post. I hope you find value in this experiment.
Statement of Problem
It is a well known fact that the combination of high levels of sodium and sulfate in brewing water will result in a harsh bitterness in a beer. According to John Palmer, it is desirable to keep either one of these minerals as low as possible, preferably the sodium. The extract brewer is faced with a handicap unless they use water nearly void of all minerals in their beer (i.e. distilled, de-ionized, or put through reverse osmosis). If an extract brewer uses their tap or well water for beer, the total minerals in the final product come from both the water used to make the extract, and the water out of their tap or well. If this sum includes high levels of sodium in combination with sulfate, the beer will have a harsh bitterness.
This was observed in one of my extract brews where I used one gallon of distilled water and the remaining water was from my tap. The initial taste of the beer was great, but it had a harsh bitterness that lasted for many minutes. Upon investigation, I realized that my tap water is high in sodium and sulfate content, 200ppm and 160ppm respectively. Since I was using extract, the minerals in the water used to make the extract also ended up in the final beer. The sum of the sodium and sulfate in both the extract water and my tap water put the harsh bitterness over the top and made the beer unpleasant.
Hypothesis
A simple solution to the problem is to use all distilled water when using extract. This will minimize the amount of both sodium and sulfate in the beer. An extract beer brewed with distilled water will be much less harsh than a beer brewed using my tap water.
Recipe
This single recipe represents two different beers. Both beers are identical to each other except for the water used.
Beer:
Sodium/Sulfate Experiment
Type: Extract
Size: ~.5 gallons
Color: 7 HCU (~6 SRM)
Bitterness: 33 IBU
OG: 1.045
FG: Not Measured (batch too small)
Alcohol: Not Measured (batch too small)
Water:
One beer was made with tap water and 1/8 tsp. gypsum creating the following concentration of minerals:
Calcium - 41ppm
Magnesium - 3ppm
Sodium - 199ppm
*Chloride - 6ppm
Sulfate - 236ppm
The final beer will include the minerals above plus the minerals used in the water to make the extract. It is expected that the combined sodium and sulfate concentrations are extremely high.
*Note the chloride/sulfate ratio of my tap water is extremely weighted to the sulfate side. I have no way of knowing what the mineral content is in the extract I used. I will be able to adjust for the low chloride level by adding calcium chloride to the beer during tasting. If the harsh bitterness is due to the combination of high levels of sodium and sulfate, then adding chloride will not get rid of the harsh bitterness.
The other beer was made with all distilled water, which theoretically has no mineral content. This allows the beer to be made with only the mineral content of the extract.
Boil:
60 minutes
SG: 1.022
1 gallons
8 oz. Amber dry malt extract
Hops:
5g Hallertauer (3.9% AA, 60 min.)
Yeast:
Safale US-05 was used. It was re-hydrated in 1/2 cup distilled water at 80°F. The yeast was allowed to hydrate for 20 minutes and then aerated for the following 30 minutes by swirling occasionally. Each beer received 1 tbsp. of the yeast slurry, equaling approximately 28 billion cells. The yeast was pitched with the worts at ~70°F.
Log:
Brew Date: 7/19/2009
Fermented in 64 oz. growlers at 64°F for 1 month.
Results
The two worts were sampled prior to pitching the yeast. A comparison is shown below.