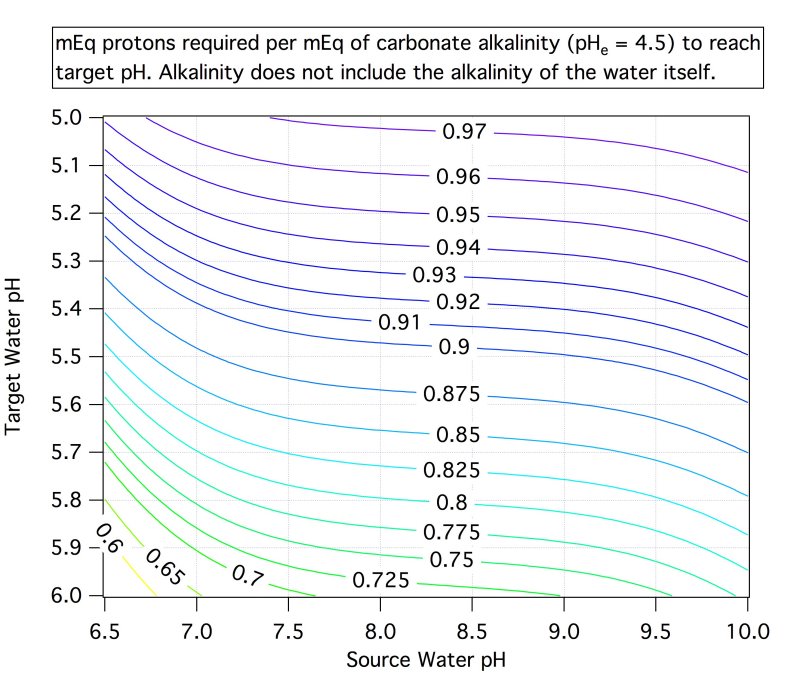
RO, distilled, and rain water are examples of low alkalinity water. Brewing with low alkalinity water is almost always a desirable thing. But, there are some mash grists that demand a little more alkalinity to keep the mash pH from dropping too low. Increasing percentages of roasted and crystal malt and grain make it more likely that alkalinity will be needed to moderate those more acidic grains.
An option for reducing the impact of those acidic grains in the mash is to steep them separately or add them at the latter part of the mash. These are workable alternatives.
Adding alkalinity to mash water can be problematic. Chalk is an unreliable alkalinity provider since you have to properly dissolve it with an acid to achieve its full alkalinity potential. Baking soda use is limited by the fact that you're adding sodium to your water and there are definite flavor impacts when added in excess. Pickling lime is a good alternative in that it adds calcium and dissolves fully, but it is a strong caustic and demands careful measurement and dosing.
It has come to my attention that there may be another option for reducing the need for alkalinity in mashing. We know that adding calcium and magnesium to the mash decreases the Relative Alkalinity (RA) of the mash water. We also know that we want a minimum concentration of calcium in our wort to promote yeast health and beer clarification.
My suggestion is that in the case of mashing with low alkalinity water, reserve any calcium or magnesium salt addition from the mash and add that directly to the kettle. In this way, the alkalinity deficiency of the mash water is not made worse by adding those salts to the mash.
This is especially true when working with a highly mineralized water profiles like Burton, Dortmond, or a Pale Ale water. For those waters, the desired sulfate or chloride concentration is often added with calcium or magnesium cations. These water profiles end up needing more alkalinity due to the elevated Ca and/or Mg and the resulting reduction in RA.
So when a program like Bru'n Water indicates that more alkalinity is probably going to be needed, try a recalculation of the mash pH by taking out the calcium and magnesium salt additions. If the mash pH prediction increases into a more desirable range, just reserve those original Ca and Mg mineral additions from the mash and put them directly into the kettle when you boil.
Enjoy!
An option for reducing the impact of those acidic grains in the mash is to steep them separately or add them at the latter part of the mash. These are workable alternatives.
Adding alkalinity to mash water can be problematic. Chalk is an unreliable alkalinity provider since you have to properly dissolve it with an acid to achieve its full alkalinity potential. Baking soda use is limited by the fact that you're adding sodium to your water and there are definite flavor impacts when added in excess. Pickling lime is a good alternative in that it adds calcium and dissolves fully, but it is a strong caustic and demands careful measurement and dosing.
It has come to my attention that there may be another option for reducing the need for alkalinity in mashing. We know that adding calcium and magnesium to the mash decreases the Relative Alkalinity (RA) of the mash water. We also know that we want a minimum concentration of calcium in our wort to promote yeast health and beer clarification.
My suggestion is that in the case of mashing with low alkalinity water, reserve any calcium or magnesium salt addition from the mash and add that directly to the kettle. In this way, the alkalinity deficiency of the mash water is not made worse by adding those salts to the mash.
This is especially true when working with a highly mineralized water profiles like Burton, Dortmond, or a Pale Ale water. For those waters, the desired sulfate or chloride concentration is often added with calcium or magnesium cations. These water profiles end up needing more alkalinity due to the elevated Ca and/or Mg and the resulting reduction in RA.
So when a program like Bru'n Water indicates that more alkalinity is probably going to be needed, try a recalculation of the mash pH by taking out the calcium and magnesium salt additions. If the mash pH prediction increases into a more desirable range, just reserve those original Ca and Mg mineral additions from the mash and put them directly into the kettle when you boil.
Enjoy!