So i'm reviving this once revived old thread because I think we can do much better.
I don't think there is a need, yet, for an experiment as this can all be proven mathematically, and any and all propaganda or articles without mathematical justification can be ignored. In particular, the wikipedia article for partial pressure
https://en.wikipedia.org/wiki/Partial_pressure
has most of the story.
The information needed is:
1) The properties by which nitrogen dissolves in a liquid is known (googlable), and quite negligible unless under extreeeeeeemly high pressures. There is a table on the wiki page for Henry's law that shows this. Co2 has medium solubility, which is why we can use it to make fizz, and nitrogen has very poor solubility.
Pure Speculation: diffusion may speed this up, just like using a stone to aerate your beer, or speed up force carbonation. I suspect this is how small amounts of nitrogen are dissolved into guinness at the factory, as claimed by the propaganda. This type of proprietary knowledge could be verified by an experiment. I liked the pH suggestion in a previous post.
2) The properties by which CO2 dissolve in a liquid are known, and most of us use some type of calculator for this.
3) The properties by which CO2 dissolves in nitrogen (and vice versa) are also known. In particular, co2 readily and easily dissolves in nitrogen
4) From the wiki: "Gases dissolve, diffuse, and react according to their partial pressures, and not according to their concentrations in gas mixtures or liquids." This behavior can be calculated using the formulae in the article.
Put it all together and I think we have the following answer. (Disclaimer: This is still speculation. I'm not a chemist..just a mathematician. I'll try to write this up as theorem and proof if I can find the time. Perhaps even a "how to" so the rest of the world can use their pocket calculators as more than a paper weight.)
1) Nitrogen's function in "beer gas" is solely to provide the added pressure to push through the restrictor plate WITHOUT over-carbonating the beer over time. This functionality can be achieved with any other gas that does not dissolve well, as pointed out by the argon experimenter posts.
2) The same results in pour can be achieved (there is a Brew Your Own article about this) by other means. Namely, crank up the co2 pressure, pour, lower the pressure, vent the keg.
3) it was claimed somewhere in this thread (EDIT:
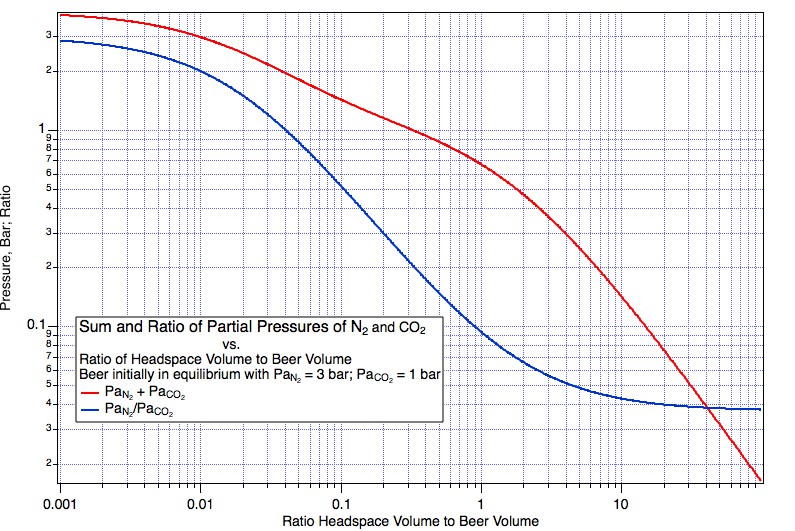
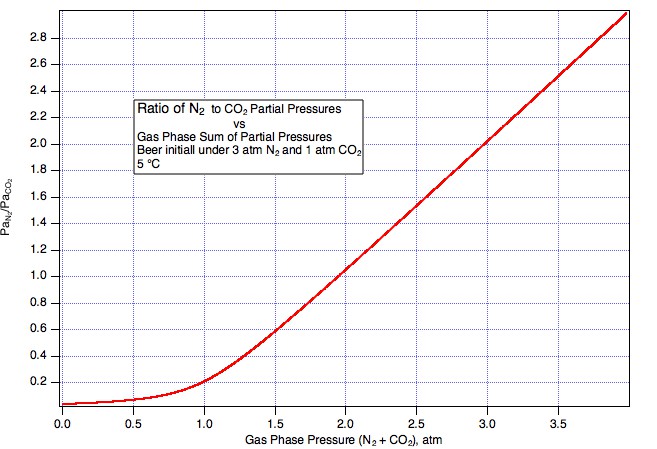
https://www.homebrewtalk.com/showthread.php?t=230229 post #5) that you could a) carbonate with co2, the b) hook up pure nitrogen and leave it with a perfect pour every time. This is a farce. Read the wiki again if you don't believe me. co2 readily dissolves in nitrogen. Thus, the pressure of pure nitrogen in the headspace does nothing to keep the co2 in solution. Only the partial pressure of co2 in the headspace does this. The partial pressure of co2 in solution will move to equilibrium with the head space, and the beer will go flat over time as co2 moves to the headspace. This is why beer gas has co2 in it, at the right partial pressure to maintain carbonation over time.
EDIT: or better yet, the first equation in the wiki shows that (in the headspace) as the volume of nitrogen increases with pints poured, the partial pressure of co2 decreases. Since co2 dissolves easily in nitrogen (this is dalton's law in the wiki), the beer goes flat.
In summary, I propose we develop a mathematical solution based on known chemistry, since none of us are privy to any proprietary information from guinness.