Well I started the Pasteurization today and I have data and plots showing the Carbonation process, as well as the Pasteurization process.
I started slightly early in the pressure rise. I was actually looking for 2.5 volumes of CO2, for a pressure of 25.3 Psi, but my basement temperature has been moving around a couple degrees as the weather is changing back and forth here in New York. This ends up changing the carbonation pressure required for a given number of volumes. I ended up doing this with 22 Psi for 2.3 volumes of CO2 as I have company coming to dinner Sunday.
Carbonation -
The carbonation process and conditioning of the bottles with a priming sugar added for 2.5 Volumes worked quite well with the plastic test bottle, with no indications of failure of either the tapped threads for the gauge or the bottle itself.
The process seems to be fairly linear over time, with a slight wander in the pressurization rate most likely due to variation in my basement temperature. Temperature will have a big impact on carbonation time.
The following data and plots show this pressurization rate.
edit: more info regarding cider prior to bottle conditioning.
Cider fermented to 1.012 using Safale S-04
Cider bottled with priming sugar added for 2.5 Volumes of CO2 on top of the finished 1.012 SG.
Cider bottle conditioned for ~ 11 days at around 62 - 66 deg
Pressure Monitored during the 11 Days, waiting for 25.3 Psi to produce 2.5 Volumes CO2.
No Cold Crash Done.
Pasteurization started at actual 22 Psi for 2.3 Volumes CO2.
Pasteurization detailed below.
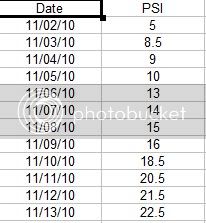
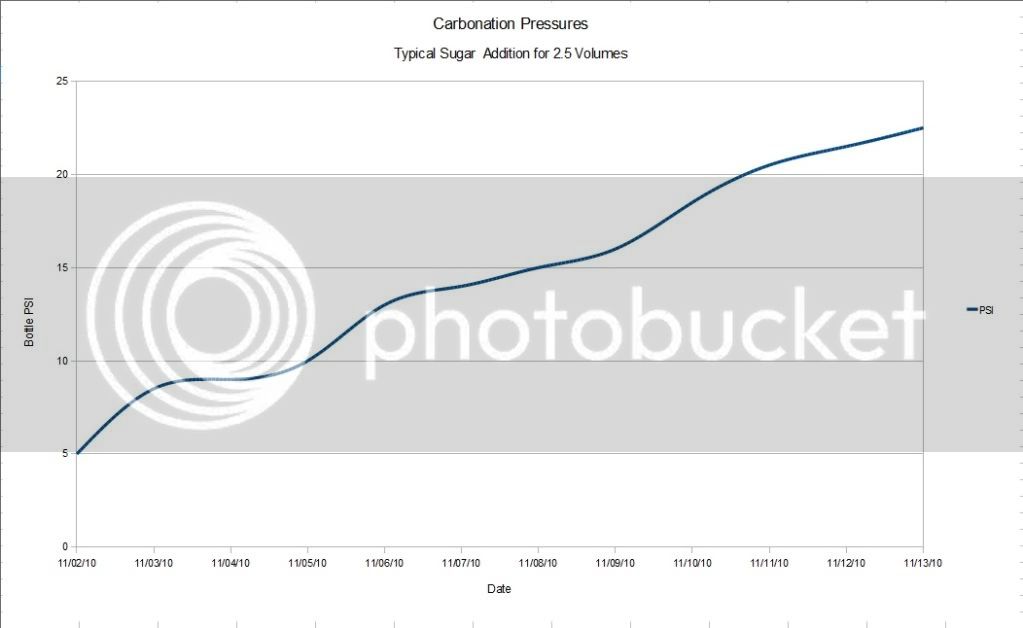
Overall, the process followed what appears to be typical when following the normal priming wait time before Pasteurization which is described quite well by Pappers sticky post.
There may be some variation in this however which could be attributed to strain of yeast and sugar content (SG) prior to adding priming sugar. This was with Safale S-04, and a final SG reading of around 1.012.
From my experience with Champagne yeast, and how vigorous that strain is, the rates could be quite different. This would support using some kind of gauging method to reach the desired pressurization value prior to Pasteurization and add a safety factor. I don't have any data using Champagne yeast yet to determine if a more aggressive yeast would change rates, however I hope to continue testing on using more than one strain to see if the rate is typical for all strains or not.
Pasteurization -
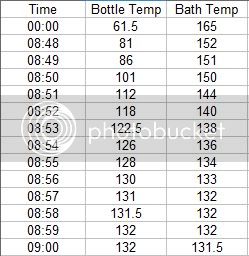
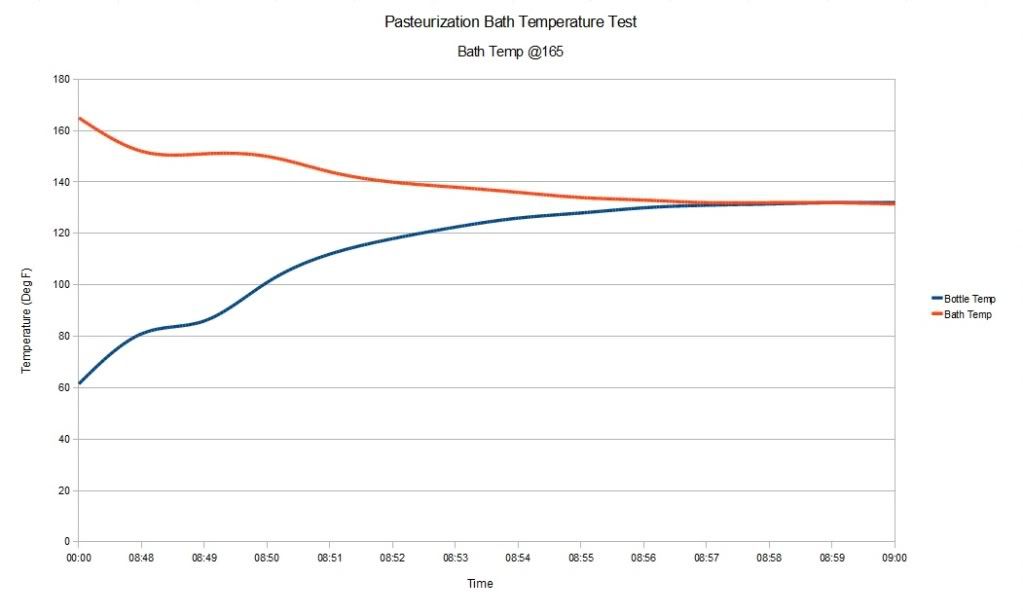
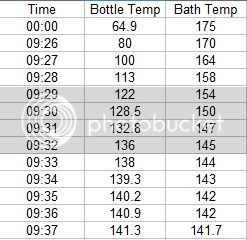
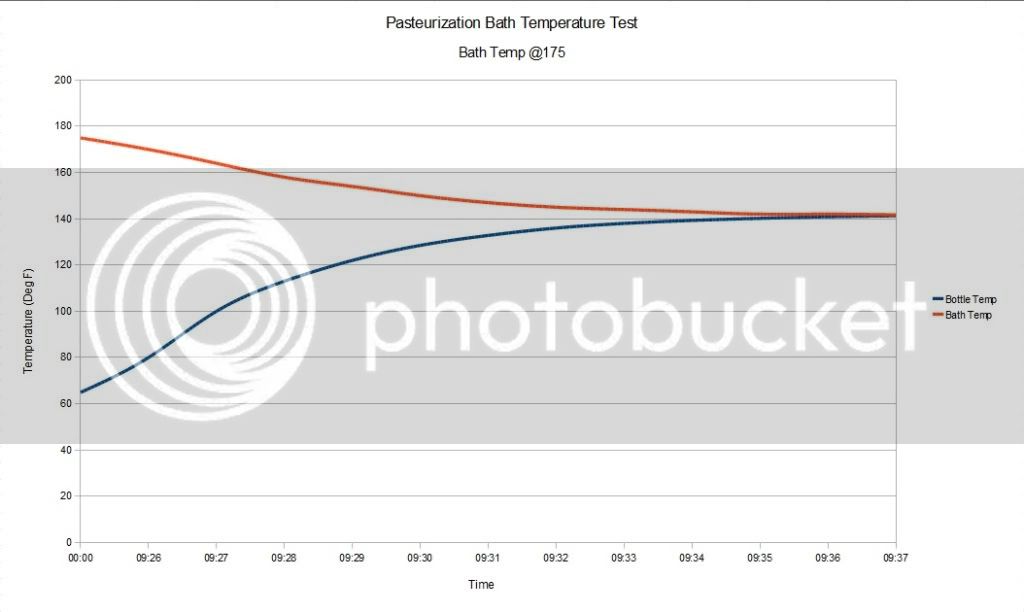
I began by putting the test bottle in a water bath of 160 degrees, with the flame off. I covered the pot with 2 towels too keep heat in as well as the water in the event the bottle burst. I recorded the pressure rise with samples at 1 minute intervals. As it turns out, the pressure inside the bottle is much higher than one might think. As you can see from the data, my 30 Psi gauge was not adequate for the Pasteurization task.
The pressure inside the bottle went way above 30 Psi. In fact the pressure did not drop and come off the 33 Psi hard stop on the gauge for almost 3 hours. As of this writing, the pressure is still above 30 Psi. The following data shows the Pasteurization process.
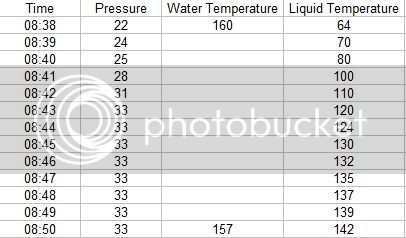
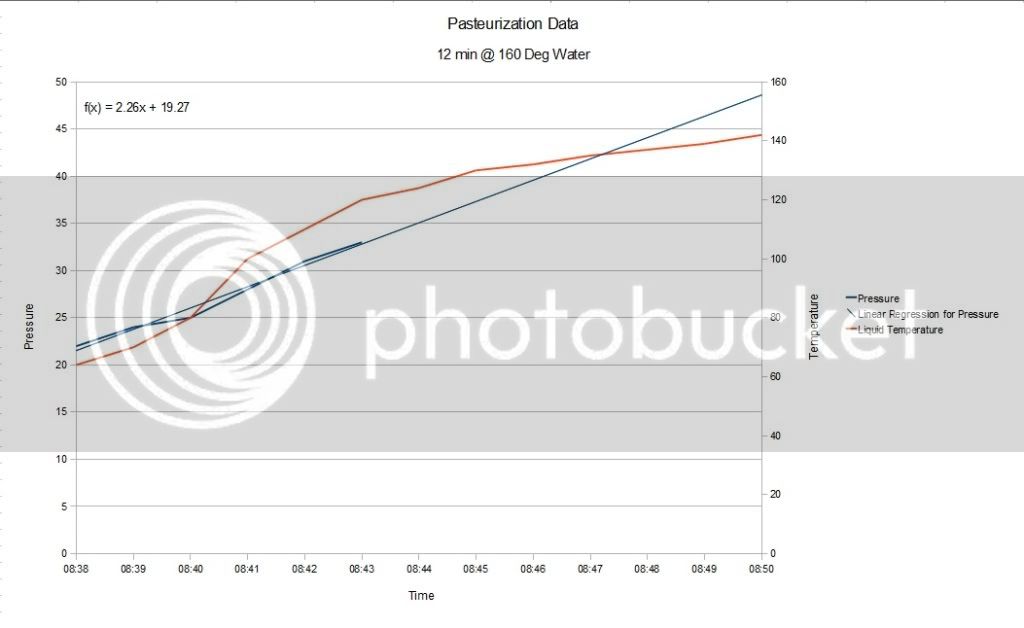
Summary -
I think that the general process of using the typical methods does work in most cases. I believe the unknowns and the method of applying Pasteurization heat with a water bath does work well, however how high a temperature of the bath will have a large impact on how high the pressure goes during that process.
Pimento has found some data regarding the temperatures that yeasts die off at, and I hope he posts that data here. However, I did use a temperature for the bath that would fall in line with his data, and would get the liquid temperature up in the 140's. If I recall correctly, his data suggests that this temperature would be fatal for the yeasts. It would also help keep the bottle pressures lower durring that process.
I used a bath temperature of 160 degrees, with a submersion time of about 12 minuets. As you can see from the data collected during the Pasteurization, the pressure climbs fairly linearly with time and with the temperature of the liquid in the container.
I wasn't able to determine the actual pressure in the bottle due to the limit of the gauge. It might make sense to use a 50 or a 100 Psi gauge for this whole process (depending on your CO2 Volumes and Pasturization Temperatures). I was able to estimate the pressure in the bottle using linear regression of the data. This of course is only a estimate, however in all likelihood a good estimate as I believe pressure of a gas increases linearly with temperature in a fixed volume.
There are a few things that may have a large impact on whether you have bottles burst during this process or not. If the bottles are submerged to a level that is either approaching or above the liquid level, the gas temperature would rise much faster than my data shows, which would cause the pressure to climb at a very rapid rate, and likely exceed what I saw or estimated. I had the water about 2/3 the way up the bottle liquid level. I think there is a fairly good chance you could blow a bottle if the water level gets a lot higher.
With all of this said, typical methods do provide a pretty good safety margin, however refining the method to provide safety and repeatability of carbonation might suggest that this method may have merit.
I think in the end, we are really force carbing the Cider like the keggers do. We raise the pressure way up, and the cider dissolves the CO2 and the pressure drops. I'll post a vid showing the Carb level I have.
Any comments or suggestions are welcome. YMMV.
Regards
Doug.
I started slightly early in the pressure rise. I was actually looking for 2.5 volumes of CO2, for a pressure of 25.3 Psi, but my basement temperature has been moving around a couple degrees as the weather is changing back and forth here in New York. This ends up changing the carbonation pressure required for a given number of volumes. I ended up doing this with 22 Psi for 2.3 volumes of CO2 as I have company coming to dinner Sunday.
Carbonation -
The carbonation process and conditioning of the bottles with a priming sugar added for 2.5 Volumes worked quite well with the plastic test bottle, with no indications of failure of either the tapped threads for the gauge or the bottle itself.
The process seems to be fairly linear over time, with a slight wander in the pressurization rate most likely due to variation in my basement temperature. Temperature will have a big impact on carbonation time.
The following data and plots show this pressurization rate.
edit: more info regarding cider prior to bottle conditioning.
Cider fermented to 1.012 using Safale S-04
Cider bottled with priming sugar added for 2.5 Volumes of CO2 on top of the finished 1.012 SG.
Cider bottle conditioned for ~ 11 days at around 62 - 66 deg
Pressure Monitored during the 11 Days, waiting for 25.3 Psi to produce 2.5 Volumes CO2.
No Cold Crash Done.
Pasteurization started at actual 22 Psi for 2.3 Volumes CO2.
Pasteurization detailed below.


Overall, the process followed what appears to be typical when following the normal priming wait time before Pasteurization which is described quite well by Pappers sticky post.
There may be some variation in this however which could be attributed to strain of yeast and sugar content (SG) prior to adding priming sugar. This was with Safale S-04, and a final SG reading of around 1.012.
From my experience with Champagne yeast, and how vigorous that strain is, the rates could be quite different. This would support using some kind of gauging method to reach the desired pressurization value prior to Pasteurization and add a safety factor. I don't have any data using Champagne yeast yet to determine if a more aggressive yeast would change rates, however I hope to continue testing on using more than one strain to see if the rate is typical for all strains or not.
Pasteurization -
I began by putting the test bottle in a water bath of 160 degrees, with the flame off. I covered the pot with 2 towels too keep heat in as well as the water in the event the bottle burst. I recorded the pressure rise with samples at 1 minute intervals. As it turns out, the pressure inside the bottle is much higher than one might think. As you can see from the data, my 30 Psi gauge was not adequate for the Pasteurization task.
The pressure inside the bottle went way above 30 Psi. In fact the pressure did not drop and come off the 33 Psi hard stop on the gauge for almost 3 hours. As of this writing, the pressure is still above 30 Psi. The following data shows the Pasteurization process.


Summary -
I think that the general process of using the typical methods does work in most cases. I believe the unknowns and the method of applying Pasteurization heat with a water bath does work well, however how high a temperature of the bath will have a large impact on how high the pressure goes during that process.
Pimento has found some data regarding the temperatures that yeasts die off at, and I hope he posts that data here. However, I did use a temperature for the bath that would fall in line with his data, and would get the liquid temperature up in the 140's. If I recall correctly, his data suggests that this temperature would be fatal for the yeasts. It would also help keep the bottle pressures lower durring that process.
I used a bath temperature of 160 degrees, with a submersion time of about 12 minuets. As you can see from the data collected during the Pasteurization, the pressure climbs fairly linearly with time and with the temperature of the liquid in the container.
I wasn't able to determine the actual pressure in the bottle due to the limit of the gauge. It might make sense to use a 50 or a 100 Psi gauge for this whole process (depending on your CO2 Volumes and Pasturization Temperatures). I was able to estimate the pressure in the bottle using linear regression of the data. This of course is only a estimate, however in all likelihood a good estimate as I believe pressure of a gas increases linearly with temperature in a fixed volume.
There are a few things that may have a large impact on whether you have bottles burst during this process or not. If the bottles are submerged to a level that is either approaching or above the liquid level, the gas temperature would rise much faster than my data shows, which would cause the pressure to climb at a very rapid rate, and likely exceed what I saw or estimated. I had the water about 2/3 the way up the bottle liquid level. I think there is a fairly good chance you could blow a bottle if the water level gets a lot higher.
With all of this said, typical methods do provide a pretty good safety margin, however refining the method to provide safety and repeatability of carbonation might suggest that this method may have merit.
I think in the end, we are really force carbing the Cider like the keggers do. We raise the pressure way up, and the cider dissolves the CO2 and the pressure drops. I'll post a vid showing the Carb level I have.
Any comments or suggestions are welcome. YMMV.
Regards
Doug.