You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Old Ale - Water Profile
- Thread starter RustyHorn
- Start date

Help Support Homebrew Talk - Beer, Wine, Mead, & Cider Brewing Discussion Forum:
This site may earn a commission from merchant affiliate
links, including eBay, Amazon, and others.
Simonh82
Well-Known Member
- Joined
- Dec 4, 2015
- Messages
- 85
- Reaction score
- 10
I would have thought you would want a more balanced water profile, with less sulphate than a Burton water profile. Old ale favours a malty rounded taste which would require more chlorine and less sulphate.
As a guess I'd go something like Ca 100ppm, Mg 5ppm, Na 25ppm, Su 50ppm, Cl 50ppm, Bicarbonate 120ppm. I'd try to keep the sulphate and chloride levels balanced.
As a guess I'd go something like Ca 100ppm, Mg 5ppm, Na 25ppm, Su 50ppm, Cl 50ppm, Bicarbonate 120ppm. I'd try to keep the sulphate and chloride levels balanced.
I would have thought you would want a more balanced water profile, with less sulphate than a Burton water profile. Old ale favours a malty rounded taste which would require more chlorine and less sulphate.
As a guess I'd go something like Ca 100ppm, Mg 5ppm, Na 25ppm, Su 50ppm, Cl 50ppm, Bicarbonate 120ppm. I'd try to keep the sulphate and chloride levels balanced.
Totally untrue. Before Burton earned its fame for PA and IPA, it was known for Burton Ale which was a big malty beer.
The problem is that the lore that sulfate makes beer 'bitter' is false. Sulfate makes a beer's finish drier...and when a beer has much bittering and hop flavor, it comes through to the drinker stronger. But, it doesn't make the beer more bitter. If there isn't much bittering in the beer, the effect of sulfate is just its drying of the finish. For a big, malty beer that could potentially be cloying, sulfate can help. I brew my Scottish beers with about 75 ppm sulfate and they are pleasingly malty and finish acceptably dry.
While I won't recommend going all the way to a Burton profile, I will say that sulfate does have a place in malty beer brewing.
Totally untrue. Before Burton earned its fame for PA and IPA, it was known for Burton Ale which was a big malty beer.
The problem is that the lore that sulfate makes beer 'bitter' is false. Sulfate makes a beer's finish drier...and when a beer has much bittering and hop flavor, it comes through to the drinker stronger. But, it doesn't make the beer more bitter. If there isn't much bittering in the beer, the effect of sulfate is just its drying of the finish. For a big, malty beer that could potentially be cloying, sulfate can help. I brew my Scottish beers with about 75 ppm sulfate and they are pleasingly malty and finish acceptably dry.
While I won't recommend going all the way to a Burton profile, I will say that sulfate does have a place in malty beer brewing.
So what would you recommend?
meatcleaver
Well-Known Member
I haven't gotten into water chemistry yet but from reading about the different profiles on brewers friend it looks like the Edinburgh profile is good for malty beers in general.
do you have a particular recipe in mind? I've been wanting to brew an old ale also & am leaning toward an Avery old jubilation clone. I freaking love that beer...
do you have a particular recipe in mind? I've been wanting to brew an old ale also & am leaning toward an Avery old jubilation clone. I freaking love that beer...
I haven't gotten into water chemistry yet but from reading about the different profiles on brewers friend it looks like the Edinburgh profile is good for malty beers in general.
do you have a particular recipe in mind? I've been wanting to brew an old ale also & am leaning toward an Avery old jubilation clone. I freaking love that beer...
It's a recipe I designed myself. I'd give you it but it's not exactly tried and tested!
Resurrecting this thread to ask: Can anyone suggest a target water profile for brewing an Old Ale?
From a variety of sources I've come up with Ca 60 ppm, Mg 10 ppm, Na 20 ppm, SO4 100 ppm, Cl 50ppm, HCO3 70 ppm. For what it's worth I'll be happier if the beer comes out too bitter than if it comes out too sweet.
I'm new at adjusting mineral content, so any insights would be appreciated.
From a variety of sources I've come up with Ca 60 ppm, Mg 10 ppm, Na 20 ppm, SO4 100 ppm, Cl 50ppm, HCO3 70 ppm. For what it's worth I'll be happier if the beer comes out too bitter than if it comes out too sweet.
I'm new at adjusting mineral content, so any insights would be appreciated.
That appears to be a decent profile for a high gravity beer. Sulfate does not make beer bitter, it makes the finish drier. In a potentially big, malty beer, helping the finish to dry out is a good thing. The worst thing you could do is boost chloride and reduce sulfate in a big beer like that.
Thanks for the lightning-fast reply! Dry sounds good. I'll try this and report back.
Totally unrelated, but here's a beginner's question: can anybody tell me where the extra cations are? I count 160 ppm of positive charge and 320 of negative. Or are ppm by mass and not number?
Totally unrelated, but here's a beginner's question: can anybody tell me where the extra cations are? I count 160 ppm of positive charge and 320 of negative. Or are ppm by mass and not number?
smata67
Well-Known Member
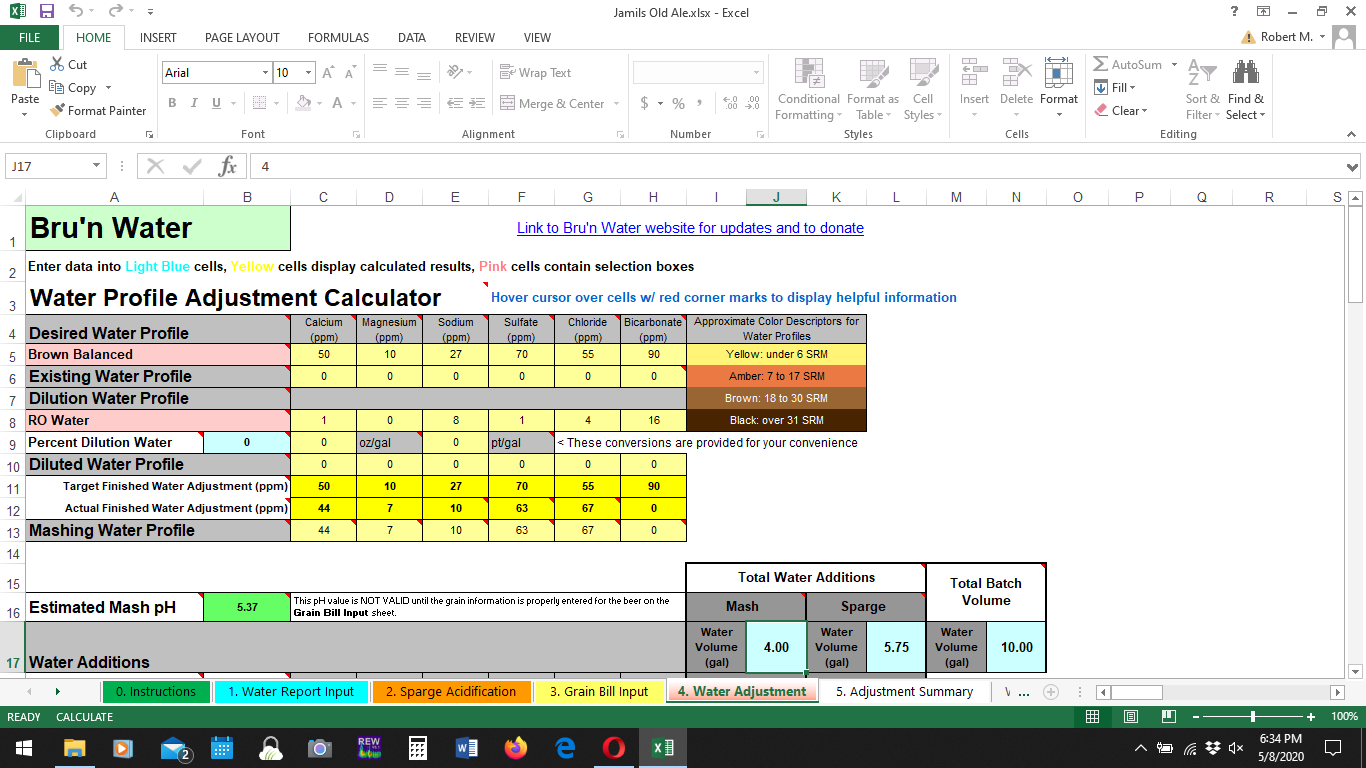
I used the Bru'n Water Brown Balanced profile for my currently conditioning Old Ale. Any comments? Did I make a good choice?



VikeMan
It ain't all burritos and strippers, my friend.
- Joined
- Aug 24, 2010
- Messages
- 5,851
- Reaction score
- 5,968
Totally unrelated, but here's a beginner's question: can anybody tell me where the extra cations are? I count 160 ppm of positive charge and 320 of negative. Or are ppm by mass and not number?
I haven't checked the math on that profile, but you do need to adjust for the weight of each ion, i.e. it's the summed charges that must balance, not the weights of the ions carrying the charges.
Similar threads
- Replies
- 5
- Views
- 399
- Replies
- 5
- Views
- 923

