markcurling
Well-Known Member
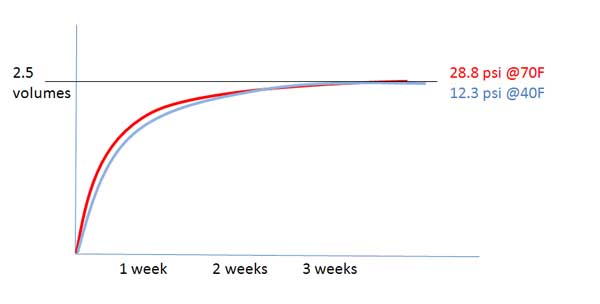
we're just arguing semantics at this point, and I am no chemist, but, since total pressure is directly proportional to temperature and partial pressures are defined by the total pressure, doesn't lowering the temperature then also lower the partial pressures?
(Or am I erroneously using ideal gas laws to talk about aqueous solutions?)
Um, possibly, I'm a bit too rusty to start delving into these realms! My instinct says that using ideal gas laws you are probably lowering the pressure of the whole closed system (both beer and headspace), however we are interested in the relative changes between the two phases inside the closed system, so there is an equilibrium shift due to the changing solubility of CO2 in water with temperature. But I don't know whether that agrees or disagrees with your point! Unfortunately I need to do something productive now, but I'll certainly check back on this thread a bit later!!